Kumar Sidhartha K Varadwaj

He holds the position of Reader at Department of Chemistry. He has more than 10 years of teaching and research experience in the extended field of materials chemistry. He has 25 publications in peer review journals to his credit. He completed Ph.D. on 2007 from IIT Kharagpur. The thesis work was based on studies of superparamagnetic particles. His postdoctoral work at KAIST, South Korea consisted of vapor phase deposition of silicide nanowires, their ensemble magnetic property and single nanowire magnetoresistance. He further moved to Research centre for integrated quantum electronics (RCIQE) at Hokkaido University, Japan where he worked on semiconductor nanowire growth on patterned structure and single nanowire lasers. He has special interest in popularisation of science through community outreach programs. His current research interest is focused on designing heterostructure materials for energy related electrocatalytic processes. Understanding the descriptors that control the activity of the catalysts. Development of electrochemical biosensors.
Selected Publications
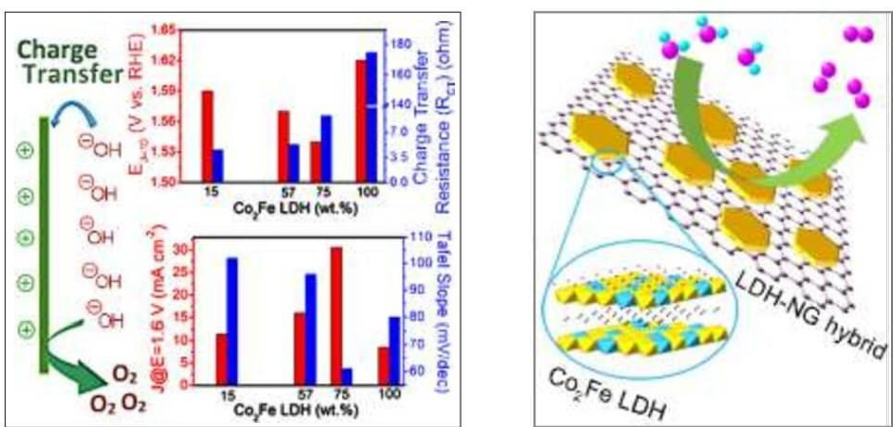
- B. D. Mohapatra, I. Hota, S. P. Mantry, N. Behera, P. Parhi and K. S. K. Varadwaj, Understanding the oxygen evolution activity trend in Co2FeLDH-N doped graphene hybrids, Electrochem. Sci. Adv. 2020, doi:10.1002/elsa.202000009.
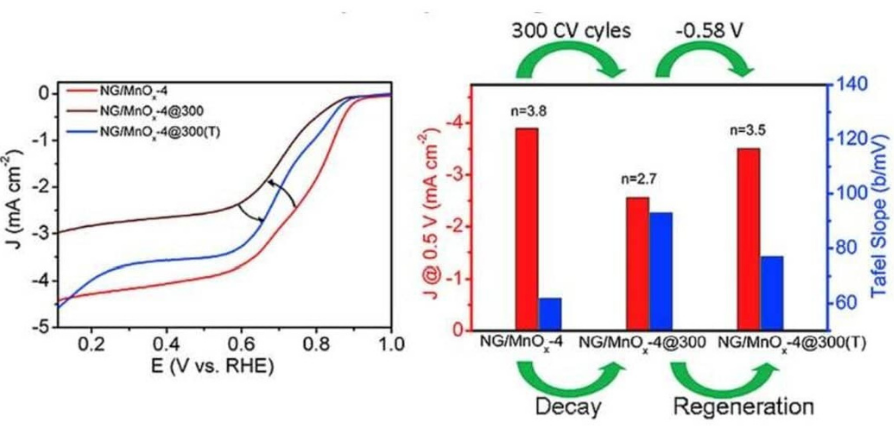
- S. P. Mantry, B. D. Mohapatra, N. Behera, P. Mishra, P. Parhi and K. S. K. Varadwaj, Potentiostatic Regeneration of oxygen reduction activity in MnOx@ Graphene hybrid nanostructures, Electrochimica Acta, 2019, 325, 134947.
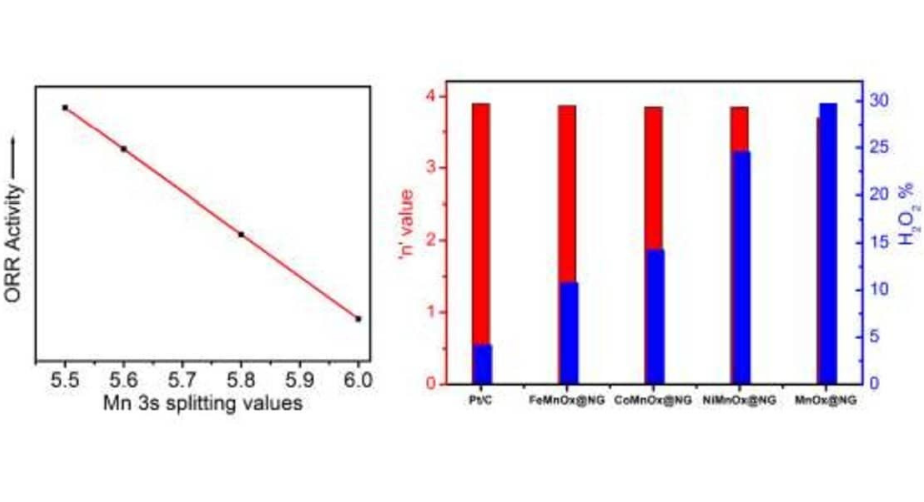
- N. Behera, S. P. Mantry, B. D. Mohapatra, R. K. Behera and K. S. K. Varadwaj, Functional molecule guided evolution of MnOx nanostructure patterns on N-graphene and their oxygen reduction activity, RSC Advances, 2019, 9, 27945–27952.
- B. D. Mohapatra, S. P. Mantry, N. Behera, B. Behera, S. Rath and K. S. K. Varadwaj, Stimulation of Electrocatalytic Oxygen Reduction Activity on Nitrogen doped Graphene through Noncovalent Molecular Functionalisation, Chem. Commun. 2016, 52, 10385-10388.
- K. S. K. Varadwaj, K. Seo, J. In, P. Mohanty, B. Kim, Phase controlled growth of metastable Fe5Si3 nanowires by a vapor transport Method, J. Am. Chem. Soc. 2007, 129, 8594-8599.

A transformative learning community to realise the dreams, spread the knowledge and create global leaders.