Founded with the Vision to spread the light of learning, and guided by the motto Jnanameva Shakti (Knowledge is power), Ravenshaw University aims to preserve its position as the “temple of learning”, with a deep knowing that the phrase exists in the vernacular for a reason. The university envisions itself as the one-stop destination for higher education for students from diverse social and economic backgrounds, and will help students to steer their passions towards making the world a better place.
With the aforesaid vision, the University has adopted three main principles – coordination, hierarchy and functional differentiation – to maintain unified goals and vision. With efficient academic governance and quality infrastructure the university targets to produce human resource commensurate with the progress of knowledge at international level. The university realizes the demand of quality higher education and requirement of connectivity between the classroom education and the societal application. It visions to initiate future programmes to impart skills, as demanded by the society and the stake holders and training of applicability.
Ravenshaw is the oldest institution of higher education in the state of Odisha and one of the oldest institutions of higher education in India. It was founded in 1868 with intermediate level (F. A.) classes at the Cuttack Zilla (district) School (now Ravenshaw Collegiate School). The high school, which was also known as the Cuttack High School, was elevated to a first grade college in 1876. It became widely known as Cuttack College until it was rechristened Ravenshaw College as a tribute to T. E. Ravenshaw, who was the Commissioner of Cuttack Division and had played a key role in its creation. The office of Ravenshaw College and Ravenshaw Collegiate School was separated in 1904.












Innovation Center
Holds over 1.8 Lakh resources
Sponsored research projects
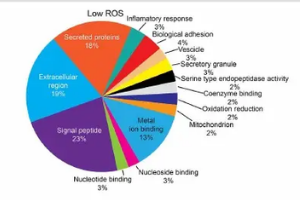
Proteomic signatures of human seminal plasma as a function of ROS gradient
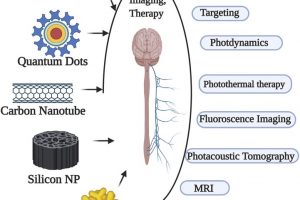
Impact of Nanoparticles on Neural Dysfunctions and Strategies to Reduce Toxicity
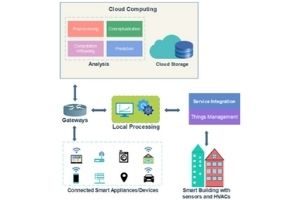
Energy Optimization & Data Reduction for IoT-Based Indoor Environments
Willingness to Pay for Rainfall Insurance in Rural India

Ravenshaw University is hosting Odisha Inter University Cultural & Literary Meet-2026, 28th-31st March 2026. 17 universities, 600 students, 22 events.
28 March 2026

Coordinated the event Echoes in Motion - Short film competition with esteemed judges Shri Bapu Lenka, Shri Basanta Sahu and Shri Asish Gadanayak. It was great to watch all the 16 entries with diverse themes with strong messages to our society.
28 March 2026

Placement season kicks off... Current stats: 400 placed graduates, 8 departments and 15 companies.
14 June 2024










