Sabita Nayak

Dr. Sabita Nayak currently an Assistant Professor at Department of Chemistry. She has more than 11 years of teaching and research experience in the extended field of organic synthesis and medicinal chemistry research. She has 36 publications in peer reviewed journals to her credit. She completed Ph.D. on 2008 from NCL, Pune under the supervision of Dr. Mukund Gurjar.. The thesis work was based on “Synthetic studies toward the total synthesis of Eunicin, Palau’amide and Asimitrin”. After completion her Ph.D., she was worked as a Research Associate in Chembiotek, Pharma- Ltd, Pune. Her postdoctoral work at University of Southwestern Medical Research Center, Dallas, Texas, USA consisted of medicinal chemistry research. She has special interest in natural product synthesis, hybrid natural product synthesis, medicinal chemistry, Carbohydrate Chemistry and 1,3 dipolar cycloaddition reaction.
Selected Publications
- D. R. Mishra, S. Nayak, B. P. Raiguru, S. Mohapatra, M. B. Podh, C. R. Sahoo, R. N. Padhy, Synthesis of (4S)-4-C-spiro-glycosyl-chromeno-[3,4-d][1,2,3]triazoles: Biological evaluation and molecular docking investigation, Journal of Heterocyclic Chemistry, 2021 , 58 ,111–126
- B. P. Raiguru, S. Nayak, D. R. Mishra, T. Das, S. Mohapatra, N. P. Mishra, Synthetic Applications of Cyclopropene and Cyclopropenone: Recent Progress and Developments, Asian journal of organic chemistry, 2020, 9,1088-1132
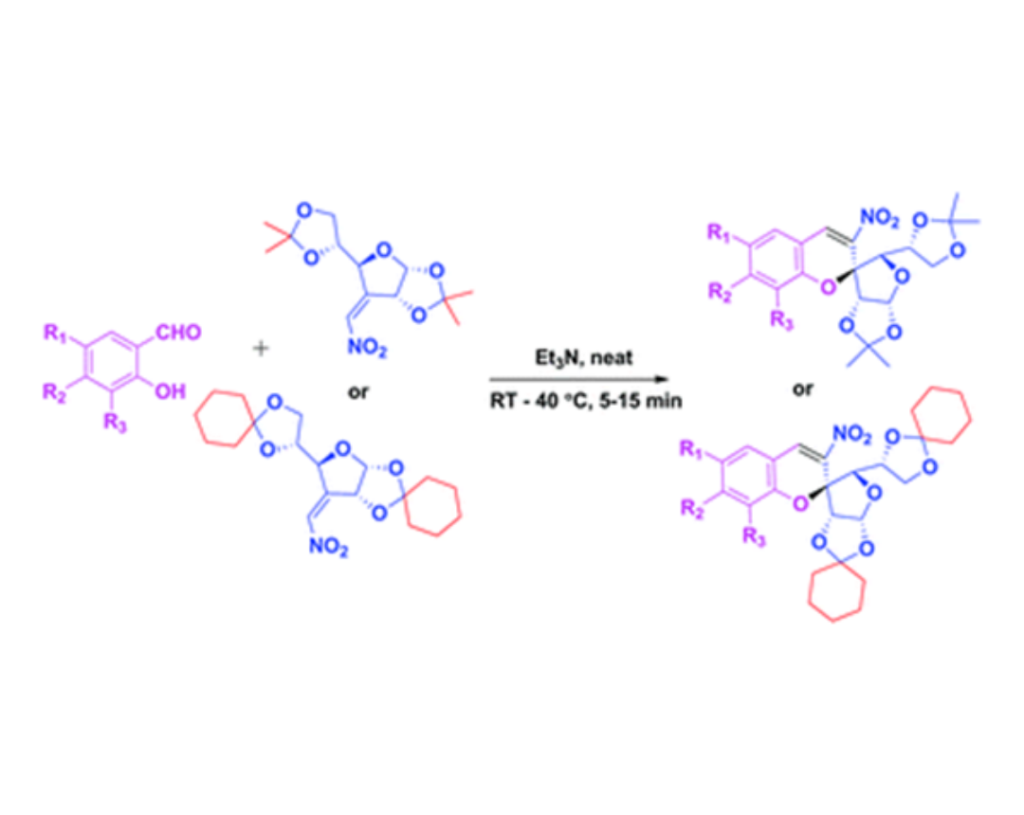
- S. Nayak, P. Panda, B. P. Raiguru, S. Mohapatra and C. S. Purohit Base mediated green synthesis of enantiopure 2-C-spiro-glycosyl-3-nitrochromenes, Org. Biomol. Chem., 2019,17, 74-82
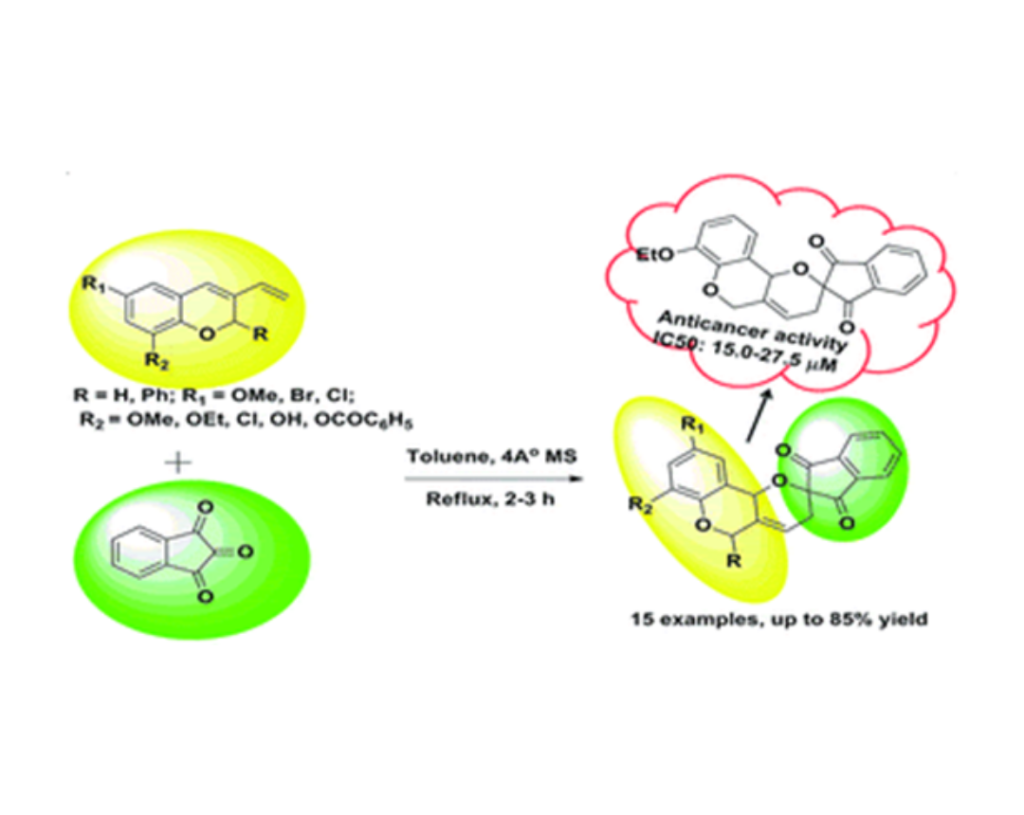
- P. Panda, S. Nayak, S. K. Sahoo, S. Mohapatra, D. Nayak, R.Pradhanb and C.N. Kundu, Diastereoselective synthesis of novel spiro indanone fused pyrano[3,2-c]chromene derivatives following hetero-Diels–Alder reaction and in vitro anticancer studies, RSC Adv., 2018,8, 16802-16814
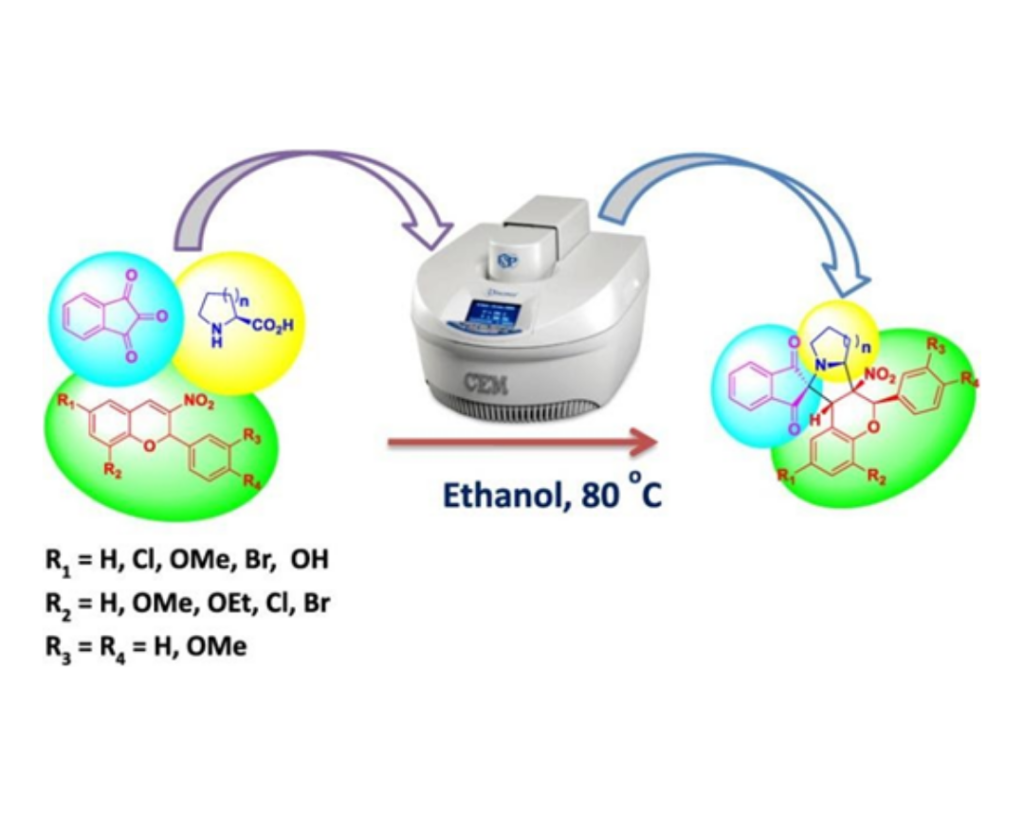
- P. Pattanaik, S. Nayak, D.R. Mishra, P. Panda, B. P. Raiguru, N. P. Mishra, S. Mohapatra, N.A. Mallampudi, C. S.Purohit. One pot, three component 1,3 dipolar cycloaddition: Regio and diastereoselective synthesis of spiropyrrolidinyl indenoquinoxaline derivatives, Tetrahedron Letters, 2018, 59, 2688-2694.

A transformative learning community to realise the dreams, spread the knowledge and create global leaders.